
Finally, all alternatives are ranked by their relative Euclidean distance from the positive solution. It can be converted to the corresponding standard SI unit J/K-mol by multiplying its value by a factor. When the maximum entropy is reached, there will no longer be any gradient of energy that will allow any spontaneous process. The universe is an adiabatic and isolated system. The maximum entropy corresponds to the thermodynamical equilibrium. Unit of Entropy (ue, eu) has a dimension of ML 2 T -2 Q -1 N -1 where M is mass, L is length, T is time, Q is temperatur, and N is amount of substance. In isolated systems, the processes leading to an increase in entropy are spontaneous. Note that the energy-level spectrum is for the total system and not. Unit of Entropy (ue, eu) is the only unit in the category of Molar entropy in our database. Then, a distance entropy model is proposed to obtain the weights of interval efficiency. unit of thermodynamic entropy, usually denoted 'e.u.' and equal to one calorie per kelvin per mole, or 4.184 joules per kelvin per mole. The entropy units arise from the Planck constant, which relates the joule and kelvin. The free energy change combines the enthalpy change and the entropy change together. where p is the pressure and V is the volume of the gas. Unit 3 - Physical Properties & Intermolecular Interactions. Types of Entropy Entropy has many types generally, but in thermodynamics, there are two types of entropies Thermodynamic Entropy One of the two types of entropy in Thermodynamic entropy. Substituting for the definition of work for a gas. The units of entropy are JK1mol1, which means that joules of energy are unavailable per unit of heat (in Kelvin) per mol. where E is the internal energy and W is the work done by the system. 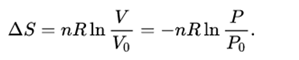
We begin by using the first law of thermodynamics: dE dQ - dW. In the proposed model, intervals of all cross-efficiency values are firstly obtained by the interval cross-efficiency method. For gases, there are two possible ways to evaluate the change in entropy. To address this issue, we propose the introduction of Shannon entropy into the cross-efficiency method. If the input or output data of DMUs are imprecise, such as the interval data, the existing methods fail to assess the efficiencies of these DMUs. The units of entropy are JK1mol1, which means that joules of energy are unavailable per unit of heat (in Kelvin) per mol. However, the existing cross-efficiency methods only evaluate the relative efficiencies of a set of DMUs with exact values of inputs and outputs. In addition, the cross-efficiency method could eliminate unrealistic weight results. The major advantage of the cross-efficiency method is that it can provide a complete ranking for all DMUs. 
It is the measure of unavailable energy in a closed thermodynamic system and. The cross-efficiency method, as a Data Envelopment Analysis (DEA) extension, calculates the cross efficiency of each decision making unit (DMU) using the weights of all decision making units (DMUs). Entropy is the measure of the thermal energy of a system per unit temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
